 Assembly and evolution of tissue-specific transcriptomic networks
Assembly and evolution of tissue-specific transcriptomic networks
Current lab members: Federica Mantica, Luis P. Iñiguez, Manuel Fernandez, Jordi Fernández-Albert, Jon Permanyer.
Former lab members: Yamile Márquez, Antonio Torres-Méndez, Demian Burguera, Reza Sodaei.
Funding: ERC Starting Grant, CRG.
Novel cell types and organismal structures in metazoans are often undergirded by complex cell type- or tissue-specific gene regulatory networks. Hence, understanding the emergence of new structures through evolution requires reconstructing the series of evolutionary steps leading to these underlying networks. However, the basic elements that form these networks are often so functionally interdependent that understanding their step-by-step emergence during evolution imposes multiple dilemmas: Did the gene network pre-exist the organismal structure? Did the targets pre-exist the tissue-specific regulators? Did the regulators ancestrally have tissue-specific expression or did they acquire it in certain lineages? Are the downstream targets conserved in the case of regulators with well-conserved expression across species?
In this research line, we try to reconstruct the stepwise assembly of various cell and tissue-specific gene and splicing networks. For this purpose, we perform comparative genomic and transcriptomic analyses of multiple homologous tissues across the relevant phylogenies. In particular, we focus on specific exon networks regulated by key splicing factors (e.g. Srrm3/4, Nova, Esrp, etc), special central components of these networks (e.g. Arc "analogs" in tetrapod and fly brains), and on the cis- and trans-regulatory architecture underlying the origin of electrocytes in electric fish.
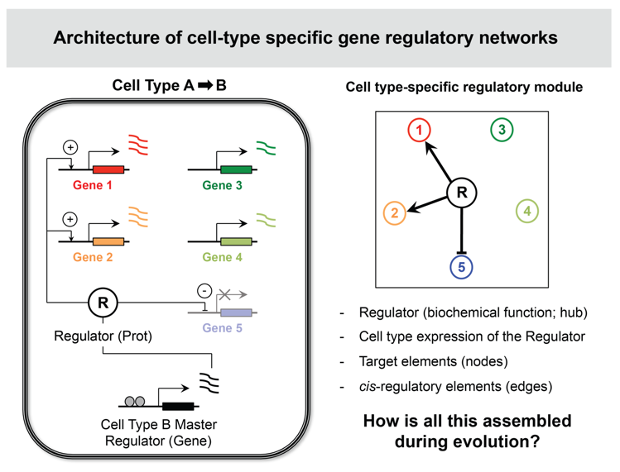
Schematic representation of a cell type-specific gene regulatory module for Cell Type B. Upon activation of a master regulator of Cell Type B, several target genes are activated (genes 1 and 2) or silenced (gene 5), which will generate the transcriptome identity of Cell Type B. Comprehending how such modules originate in evolution requires understanding how the master regulator, its cell type-specific expression and its targets and regulatory motifs all co-evolved together.
In addition to focus on individual networks of interest, in this research line we also perform broad comparative transcriptomic analysis to study tissue-specific programs across a large phylogenetic range. With a strong focus on the origin of vertebrates, we have done various comparisons between vertebrates and non-vertebrates (covering >600 million years), in contrast to the most common studies, which mostly target mammals (~100 million years). The methodological and conceptual challenges are very different and our expertise thus places us in a unique position to address key unanswered questions. With this in mind, we have generated extensive transcriptomic data to cover eight homologous tissues across twenty bilaterian species. This exceptional dataset provides us with the source to perform an unprecedented comparative investigation of the evolution of tissue-specific transcriptomes across 600 million years of evolution, focusing on gene expression, alternative splicing and other post-transcriptional regulatory layer. We hope to unveil a plethora of tissue-specific novelties across species that we will experimentally follow up.
Related publications:
- Mantica, F., Iñiguez, L.P., Marquez, Y., Permanyer, J., Torres-Mendez, A., Cruz, J., Franch-Marro, X., Tulenko, F., Burguera, D., Bertrand, S., Doyle, T., Nouzova, M., Currie, P., Noriega, F.G., Escriva, H., Arnone, M.I., Albertin, C.B., Wotton, K.R., Almudi, I., Martin, D., Irimia, M.† (2022). Pervasive evolution of tissue-specificity of ancestral genes differentially shaped vertebrates and insects. https://www.biorxiv.org/content/10.1101/2022.11.14.516384v1.
- Mantica, F., Irimia, M.†. (2022). The 3D-Evo Space: Evolution of Gene Expression and Alternative Splicing Regulation. Annu Rev Genet, 56:315-37.
- Torres-Méndez, A.†, Pop, S., Bonnal, S., Almudi, I., Roberts, R.J.V., Paolantoni, C., Alcaina, A., Avola, A., Martín-Anduaga, A., Haussmann, I.U., Morin, V., Casares, F., Soller, M., Kadener, S., Roignant, J.-Y., Prieto-Godino, L.†, Irimia, M.† (2022). Parallel evolution of a splicing program controlling neuronal excitability in flies and mammals. Sci Adv, 8:eabk0445.
- Martín, G.†, Márquez, Y., Mantica, F., Duque, P., Irimia, M.† (2021). Alternative splicing landscapes in Arabidopsis thaliana across tissues and stress conditions highlight major functional differences with animals. Genome Biol, 22:35.
- Marquez, Y.†, Mantica, F., Cozzuto, L., Burguera, D., Hermoso-Pulido, A., Ponomarenko, J., Roy, S.W., Irimia, M.† (2021). ExOrthist: a tool to infer exon orthologies at any evolutionary distance. Genome Biol, 22:239.
- Torres-Méndez, A., Bonnal, S., Marquez, Y., Roth, J., Iglesias, M., Permanyer, J., Almudí, I., O’Hanlon, D., Guitart, T., Soller, M., Gingras, A.-C., Gebauer, F., Rentzsch, F., Blencowe, B.J.B., Valcárcel, J., Irimia, M.† (2019). A novel protein domain in an ancestral splicing factor drove the evolution of neural microexons. Nature Ecol Evol, 3:691-701.
- Irimia, M.†, Maeso, I. (2019). Boosting macroevolution: genomic changes triggering qualitative expansions of regulatory potential. On: Old questions and young approaches to animal evolution. Springer-Nature.
- Marletaz, F., Firbas, P., Maeso, I., Tena, J.J., Bogdanovic, O., Perry, M., Wyatt, C.D.R., [+50 authors including Marquez, Y., Burguera, D. and Permanyer, J.], Holland, P.W.H., Escriva, H.†, Gomez-Skarmeta, J.L.†, Irimia, M.† (2018). Amphioxus functional genomics and the origins of vertebrate gene regulation. Nature, 564:64-70.
- Grau-Bove, X., Ruiz-Trillo, I.†, Irimia, M.† (2018). Origin of exon skipping-rich transcriptomes in animals driven by evolution of gene architecture. Genome Biol, 19:135.
- Burguera, D., Marquez, Y., Racioppi, C., Permanyer, J., Torres-Mendez, T., Esposito, R., Albuixech, B., Fanlo, L., D'Agostino, Y., Gohr, A., Navas-Perez, E., Riesgo, A., Cuomo, C., Benvenuto, G., Christiaen, L.A., Martí, E., D'Aniello, S., Spagnuolo, A., Ristoratore, F., Arnone, M.I.†, Garcia-Fernàndez, J.†, Irimia, M.† (2017). Evolutionary recruitment of flexible Esrp-dependent splicing programs into diverse embryonic morphogenetic processes. Nat Commun, 8:1799.
- Solana, J.*†, Irimia, M.*†, Ayoub, S., Orejuela, M.R., Zywitza, V., Jens, M., Tapial, J., Ray, D., Morris, Q.D., Hughes, T.R., Blencowe, B.J., Rajewsky, N.† (2016). Conserved functional antagonism between CELF and MBNL proteins regulates stem cell-specific alternative splicing and regeneration in planarians. Elife, 5:e16797.

